Face oil: Is it the right fit for you?
Unlock the potential of face oil for radiant, hydrated skin. Learn how omega-3 fatty acids and essential nutrients nourish and protect your skin barrier.

Unlock the potential of face oil for radiant, hydrated skin. Learn how omega-3 fatty acids and essential nutrients nourish and protect your skin barrier.

India’s cultural consciousness is embedded with pearls of heritage carried forward through generations in the form of folk art.
Health outcomes vary greatly among the approximately 1.5 million Americans who survive a traumatic brain injury each year.
For millions of adults in the United States, involuntary loss of bowel control, or fecal incontinence (FI), has a major negative influence on their quality of life and mental health.
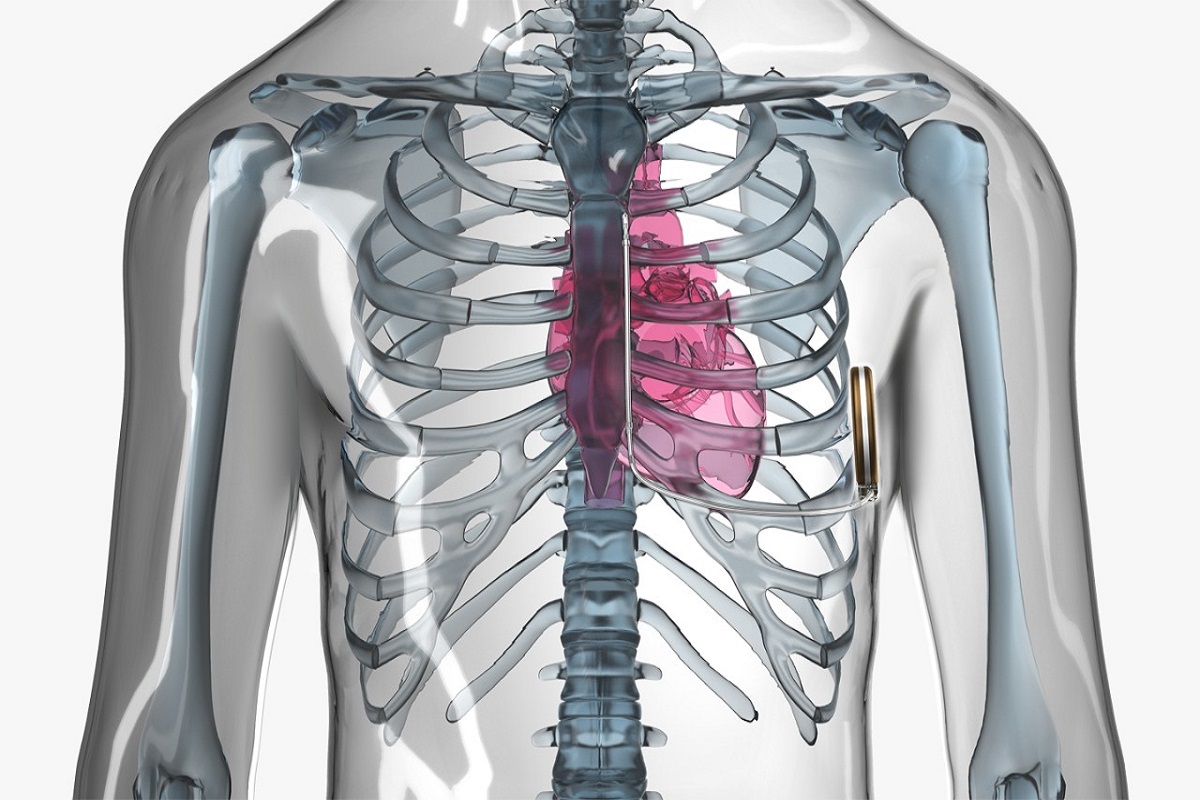
When the patient required anesthesia following successful resuscitation, midazolam increased the likelihood of adequate oxygen saturation and CO2 levels in the blood.
The Indian beauty market is estimated to reach $2.27 billion by 2028, with a CAGR of 10.91% during the forecast period (2023-2028).
Discover effective DIY Ayurvedic foot care solutions from Patanjali for revitalized, healthy feet. Explore natural remedies and products for soft, supple soles.
Discover a variety of irresistible banana recipes that are both nutritious and budget-friendly. From classic banana bread to flavorful curry and fluffy pancakes, explore new ways to enjoy this versatile fruit!
Learn about the early symptoms and management of Haemophilia, a rare genetic disorder characterized by impaired blood clotting, in this informative guide.
Whether you're sweltering in dry heat or drowning in humidity, the hills are calling to cool you down.