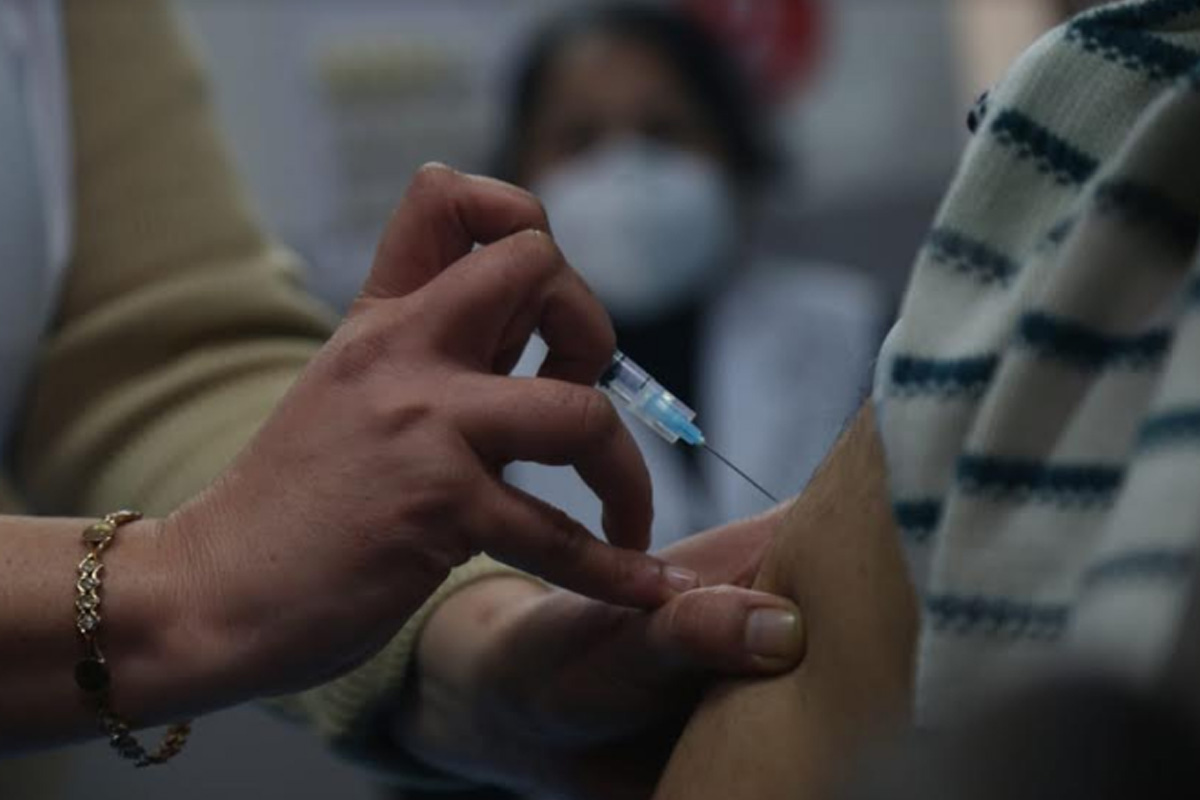
A42-year old ambulance driver from Nirmal in Telangana died within 18 hours of being vaccinated after developing chest pains though the state health department has said according to preliminary report the death was unrelated to the vaccination.
“One male healthcare worker, 42 years old, was vaccinate at about 11:30 a.m. on 19 January in Kuntala PHC, Nirmal district. At about 2:30 a.m. on 20 January he is said to have developed chest pain and was brought dead to district hospital Nirmal today around 5:30 a.m,” read the statement released by the director of public health and family welfare.
Advertisement
According to the statement, preliminary findings suggest that “the death seems to be unrelated to vaccination”.
However, according to guidelines post mortem will be conducted by a team of doctors.
The district AEFI committee which is examining the matter will submit its report to the state level committee which in turn will furnish its report to the central AEFI committee to take a view.
Telangana began the vaccination programme on 16 January and has so far more than 70 AEFI cases though this is the first death that has been reported. Some health workers in Osmania Hospital have reported fever and body aches after the jab.
Meanwhile, in a letter to Union minister for health and family welfare Harsh Vardhan KT Rama Rao iterated the request for establishment of a vaccine testing and certification laboratory in Hyderabad, similar to the Central Drug Laboratory at Kasauli in Himachal Pradesh.
Hyderabad is known as the vaccine capital of the world, manufacturing more than 6 million doses each year and contributing onethird of the global vaccines output and the Telangana industry minister pointed out the vaccines manufactured in the city are being sent to Central Drug Laboratory in Kasauli for testing and certification which due to the logistics and time involved was constraining the industry.
Since the state government has already provided land to Indian Council for Medical Research to establish the country’s largest National Animal Resource facility for biomedical research in Genome Valley Rao urged the Union Minister to expedite the project as “this facility already has a mandate for testing and certifying the drug including vaccines, medical devices and to certify them on behalf of the Government of India I would like to request that the establishment of this facility be expedited and this institution is strengthened further to conduct testing and prerelease certification of Immuno biologicals meant for human use in India.” said KT Rama Rao.
He also demanded setting up of a Government Medical Store Depot facility in the city.