Beyond the cell
Both plant and animal cells have extracellular structures an in case of animal cells, the extracellular matrix consists of collagen fibres embedded in a network of glycosaminoglycans and proteoglycans.
Secretory pathways transport molecules to the exterior of the cell.
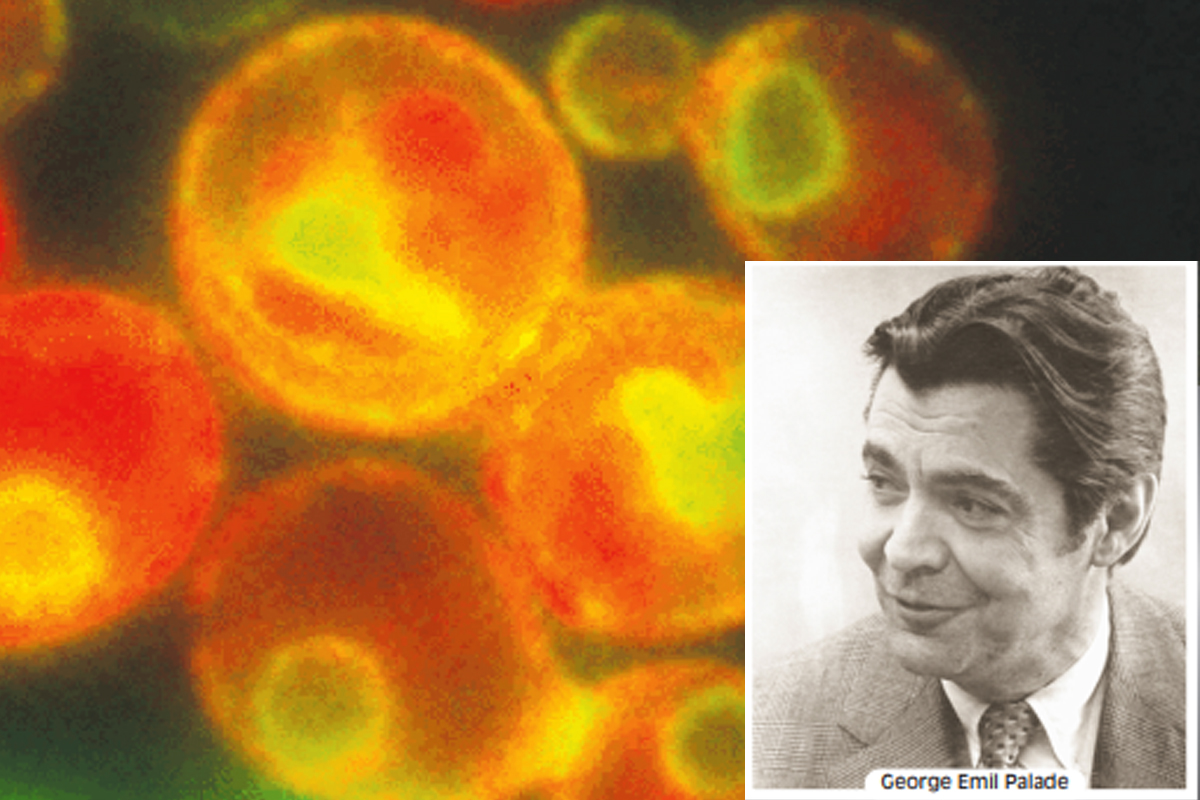
(Photo: Statesman News Service)
Integral to the vesicular traffic are secretory pathways by which proteins move from the endoplasmic reticulum through the Golgi complex to secretory vesicles and secretory granules, which then discharge their contents to the exterior of the cell. The concerted roles of the ER and the Golgi complex in secretion were first demonstrated by Nobel Laureate George Emil Palade and his colleagues, who used autoradiography to trace the fate of radioactively labelled protein through cells in the salivary gland.
The results of one of Palade’s experiments were that for the first few minutes after injection of a radioactive amino acid into rabbits, newly synthesised protein containing the radioactive amino acid was found only in the rough ER of the secretory tissue. Shortly thereafter, radioactivity began to appear in the Golgi complex, peaking at about 30-40 minutes. By 30 minutes, radioactively labelled protein could also be detected in vesicles budding from the Trans-Golgi Network, which Palade called condensing vacuoles. After 90 minutes, radioactivity began to accumulate in secretory granules, the vesicles that discharge secretory proteins to the exterior of the cell. Radioactively labelled protein eventually was released in the extracellular medium, demonstrating that the secretory granules actually release their contents beyond the plasma membrane.
Advertisement
Based on Palade’s initial experiments and numerous similar studies since then, the secretory pathways are now understood in considerable detail. Moreover, there are two different modes of secretion by eukaryotic cells. Constitutive secretion involves the continuous discharge of vesicles at the plasma membrane surface, whereas regulated secretion entails controlled rapid releases that are usually in response to an extracellular signal. After budding from the TGN, some secretory vesicles move directly to the cell surface, where they immediately fuse with the plasma membrane and release their contents. This process, which is continuous and independent of specific extracellular signals, occurs in most eukaryotic cells and is called constitutive secretion. Examples include the continuous release of mucus by cells that line your intestine and the secretion of the glycoproteins of the extracellular matrix. Constitutive secretion was once assumed to be a default pathway for proteins synthesised by ribosomes attached to the rough ER. According to this model, all proteins destined for transport from the ER to the Golgi complex must have a tag that diverts them from constitutive secretion. Unless an amino acid sequence or oligosaccharide chain identifies a protein for retention in, or transport to, a specific organelle, the protein moves through the endomembrane system and is released outside the cell by default. Support for this model came from studies in which the retrieval tags on ER-specific proteins were removed and the fates of the proteins were traced. Removal of the sequence from resident proteins of the ER generally led to secretion of the protein. More recent evidence, however, points to a variety of short amino acid sequences that identify proteins for constitutive secretion by one of several distinct pathways for this process.
Advertisement
While vesicles containing constitutively secreted proteins move continuously and directly from the TGN to the plasma membrane, secretory vesicles involved in regulated secretion accumulate in the cell and then fuse with the plasma membrane only in response to specific extracellular signals. An important example is the release of neurotransmitters. Two additional examples of regulated secretion are the release of insulin from the ? cells of the pancreatic islets of Langerhans and the release of digestive enzymes from acinar cells of the pancreas.
Regulated secretory vesicles form by budding from the TGN as immature secretory vesicles, which lose their clathrin coats and undergo a maturation process. Maturation involves concentration of the proteins — referred to as condensation — and frequently also the proteolytic processing of secretory proteins. The mature secretory vesicles then move close to the site of secretion and remain near the plasma membrane, until receiving the signal that triggers release of their content by exocytosis.
are usually quite large and contain much more highly concentrated proteins than do constitutive secretory vesicles. Such large dense vesicles are often called secretory or zymogen granules to distinguish them from other secretory vesicles. Zymogen granules are con-centrated in the region of the cell between the Golgi stacks from which they arise and the portion of the plasma membrane bordering the lumen into which the contents of the granules are eventually discharged.
The information needed to direct a protein to a regulated secretory vesicle is presumably inherent in the amino acid sequence of the protein, though the precise signals and mechanisms for sorting proteins to secretory vesicles are not clear. Current evidence suggests that high concentrations of secretory proteins in secretory granules promote the formation of large protein aggregates that exclude non-secretory proteins. This could occur in the TGN, where only aggregates would be packaged in vesicles destined for secretory granules, or it could occur in the secretory granule itself. The pH of the TGN and the secretory granule lumens may serve as a trigger favouring aggregation as material leaves the TGN. The soluble proteins that do not become part of an aggregate in the TGN or a secretory granule would be carried by transport vesicles to other locations.
Advertisement