Microscopic life: From chaos to classification
Micro-organisms constitute a very antique group of living organisms which appeared on the Earth's surface almost 3000 million years ago.
A team of scientists has published a paper recently that looks into how amino acids formed on early Earth to birth complex life.
(Photo: Statesman News Service)
The molecular basis of genetics, reproduction and evolution has enabled scientists to trace our origins back to the earliest of life forms. How the first living thing arose, to reproduce and start the process, however, has been a puzzle. The puzzle arises because the mechanics of life depend on proteins, the generation of which is the hallmark of life. Proteins, in their millions of forms, are assembled from a base-set of just 20 molecules known as amino acids. Assembling amino acids into proteins, however, consumes energy and depends on catalysts known as enzymes. Now, enzymes are proteins themselves.
How then, could these proteins, which we need for protein assembly, have been assembled in the first place? This is the “chicken and egg” problem that has troubled scientists. Pierre Canavelli, Saidul Islam and Matthew W Powner, from University College London, London, report in the journal, Nature, that they may have found the answer. The processes that underlie the evolution of species from earlier life forms unfolded when the mechanisms of reproduction and heredity were understood. A mystery that had long puzzled scientists was how the great variety of species that exist could have come about. One theory, proposed by the naturalist, Jean-Baptiste Lamarck, was of the transmutation of the species — the simplest creatures arose spontaneously and were driven, by a life force that infused them, to increasing complexity.
Advertisement
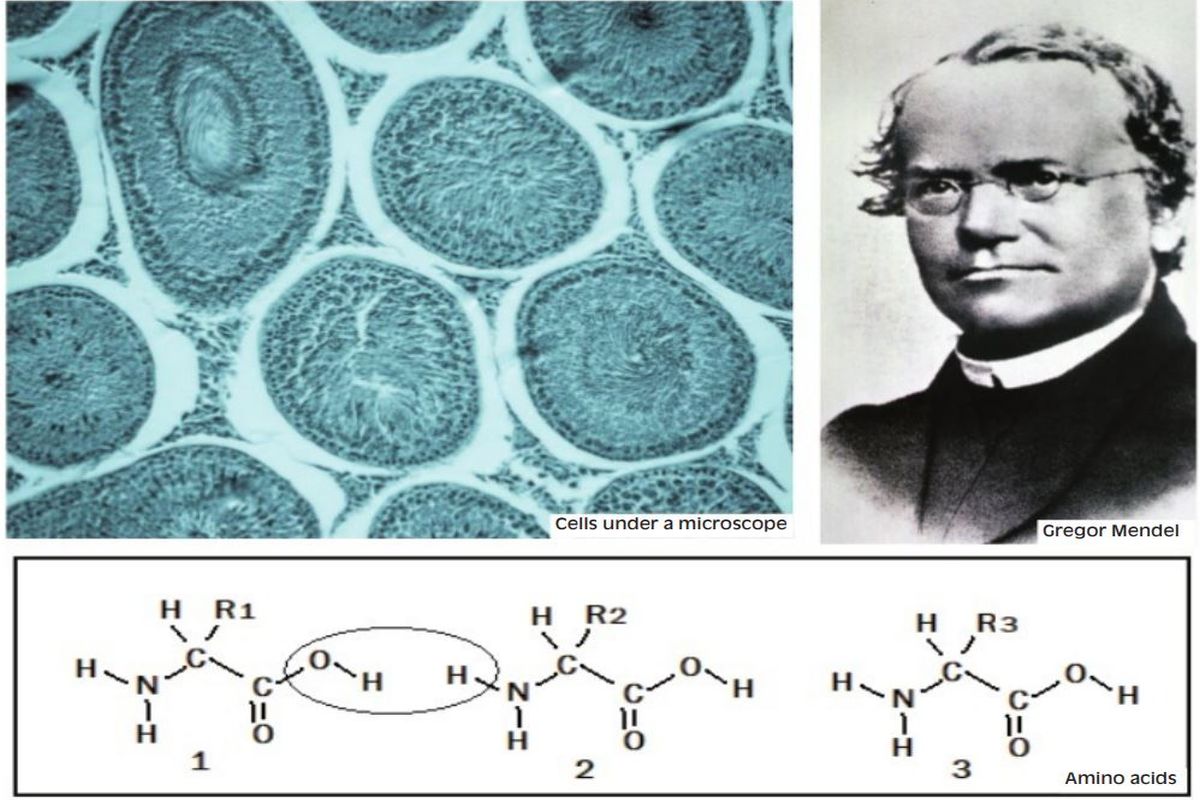
There was also the experience of controlled breeding, which could promote specific traits in domestic animals. There was no indication, however, of what the driver was in the natural world. It was Charles Darwin who realised that it was for the survival of the fittest that traits were selectively bred among living things in the wild. The work of Gregor Mendel was followed by the discovery that living cells had components that participated in reproduction and heredity. This led to the discovery that the nucleus of every cell contained a chain molecule called DNA, which programmed what proteins the cell would produce. Much of the structure of a cell is made up of proteins and virtually all actions of the cell are triggered by proteins.
Advertisement
The proteins a cell produces hence determines what a cell is — a brain cell or a liver cell, and the cell of a human, animal or plant. The DNA molecule is millions of units long and consists of segments, each of which contains the blueprint for a different protein. As the DNA can also create a duplicate of itself when the cell divides, it is a device that explains both reproduction and heredity. It could hence be conceived that the great diversity of life forms we have, could have evolved from the first, rudimentary organism, with a bare-bones DNA, which arose out of the soup of chemicals that once covered parts of the Earth.
The trouble with this picture, however, comes into view when we examine how the information in the DNA is made use of to create proteins. Each of the segments in the DNA consists of a series of triplets, or groups of three chemical forms, called “bases”, selected out of four possible kinds. These four kinds are known as A, C, G and T, and each triplet has to be made up of bases only from these four. As each of the three members of the triplet can be anyone for four alternatives, we can see the total number possible triplets is 4x4x4=64.
Hence, if each triplet is used to identify a particular molecule, 64 different molecules can be identified by the “three-character code” formed by one triplet. The 64 codes, however, are used to identify only 20 amino acids, the building blocks of proteins, by providing duplicate codes, to prevent errors, and allow for triplets to mark the “start” and the “end” of a series. And with 20 identified, a chain of triplets can stand for a great variety of sequences or amino acids, and hence for millions of different proteins. Now for how the code in the DNA results in proteins getting formed. While the DNA is inside the nucleus, the formation of proteins takes place outside the nucleus. The portion of the DNA that codes for a particular protein, as marked by the “start” and “end” triplets, is first copied, in a form that is similar to the DNA, on to a strand called the messenger RNA, or mRNA.
When the mRNA is in the outer part of the cell, other functional parts of the cell get active for the specific amino acids to line up, according to the sequence of triplet codes contained in the mRNA. With the amino acids thus arranged, the protein molecule can take form. The trouble is that it takes energy for the amino acids to pair up, with neighbours, to form a chain. The structure of amino acids, as shown in the picture, is that they all have an “NH” group at one end and an “O-H” group at the other, the difference is according to the nature of the group, “R”. Now, the “H” at the first end of one amino acid can combine with the “O-H” at the other end of the next amino acid, to form a molecule of H2O, or water, which gets released, leaving the two amino acids linked or bonded.
Now, for this energy-consuming reaction that leads to the linking of amino acids to proceed rapidly, there is a need for special molecules that can ease the process. Such catalysts and sites where amino acids need less energy to bond are provided by bodies called ribosomes, with which enzymes and other proteins are associated. Hence the question — how were the first promoters of amino acid bonding created, when their very creation would involve the formation of amino acid bonds? As the bonding of amino acids consumes energy and releases water, the presence of water can result in the breaking of the bond. The University College authors of the paper in Nature note that there is evidence that the first linked amino acid chains came into being before the first living things and played a part in the origin of life.
But there is no plausible way these chains could have formed in the watery medium of early Earth. Rather than look for a way to get amino acids to form bonds while bathed in water, the authors turned their gaze on amino nitriles — compounds that contain less “H” and “O” atoms — and combine with water, giving off energy, to form amino acids. These compounds, which are an earlier form of amino acids, were found to be capable of linking up directly to form the amino acid chain without the intermediate, “combining with water and then giving up the water” phase. The group has found that this reaction took place and the reagents necessary were available during the primitive times when the first life forms appeared. The team is now studying other ways to form amino acid chains, and the properties of chains that formed in this way, to learn more of how life, which sustains itself, was “bootstrapped” into existence.
Advertisement