BJP manifesto yet another poll gimmick: Pratibha Singh
Himachal Pradesh Congress President and MP Pratibha Singh has termed BJP’s Lok Sabha poll manifesto yet another poll ‘gimmick’.
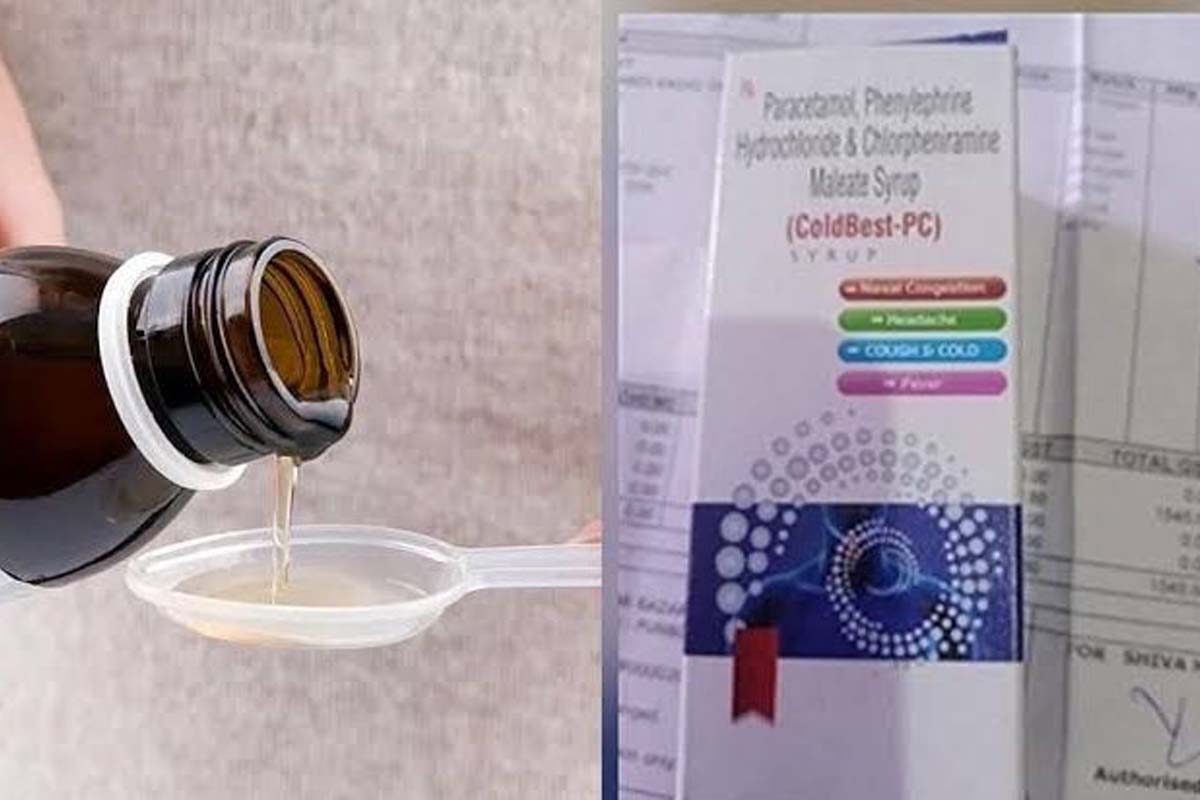
In a suo motu statement in the state Assembly, the CM said the Drug Controller of J&K had brought the matter of death of children in Udhampur area due to presence of Diethylene in Coldbest-PC cough syrup (batch no. 5201) by M/S Digital Vision Kala Amb based Pharma company to the notice of state government through an e-mail on 15 February.
[Representational Image]
Chief Minister, Jai Ram Thakur today said strict action has been taken against HP based cough syrup manufacturing company after preliminary investigation revealed that the syrup was found contaminated.
In a suo motu statement in the state Assembly, the CM said the Drug Controller of J&K had brought the matter of death of children in Udhampur area due to presence of Diethylene in Coldbest-PC cough syrup (batch no. 5201) by M/S Digital Vision Kala Amb based Pharma company to the notice of state government through an e-mail on 15 February. The CM, however, did not mention that number of deaths. (There were reports of death of 12 children in this regard).
The CM said a case was registered against the company under sections 18 (a) (1) read with 17 A of Drugs Cosmetics Act which is punishable under section 270 for at least ten years upto life imprisonment. Section 308 of Indian Penal Code (IPC) for attempt to commit culpable homicide was also added in the FIR, he added.
Advertisement
The CM said the manufacturing license of the pharma company has been suspended pending inquiry.
He said the FIR had been registered at Kala Amb police station after getting a report on March 2 from Chandigarh based Government Analyst laboratory of the samples of Coldbest-PC cough syrup taken from Haryana and Jammu-Kashmir.
The report stated that the syrup produced by the company contained over 34 percent Diethylene glycol. Thakur said the batch has been withdrawn from the market and police are investigating the matter in detail.
Advertisement