Fantasy sports revenue growing bigger thanks to IPL: Report
Gaming app platforms like Dream11 and My 11 Circle have shown an impressive surge in both daily active cash users and revenue per person.

Gaming app platforms like Dream11 and My 11 Circle have shown an impressive surge in both daily active cash users and revenue per person.

His comments come amid recent attacks on Israel by Iran, reportedly in retaliation to the attack on their embassy.
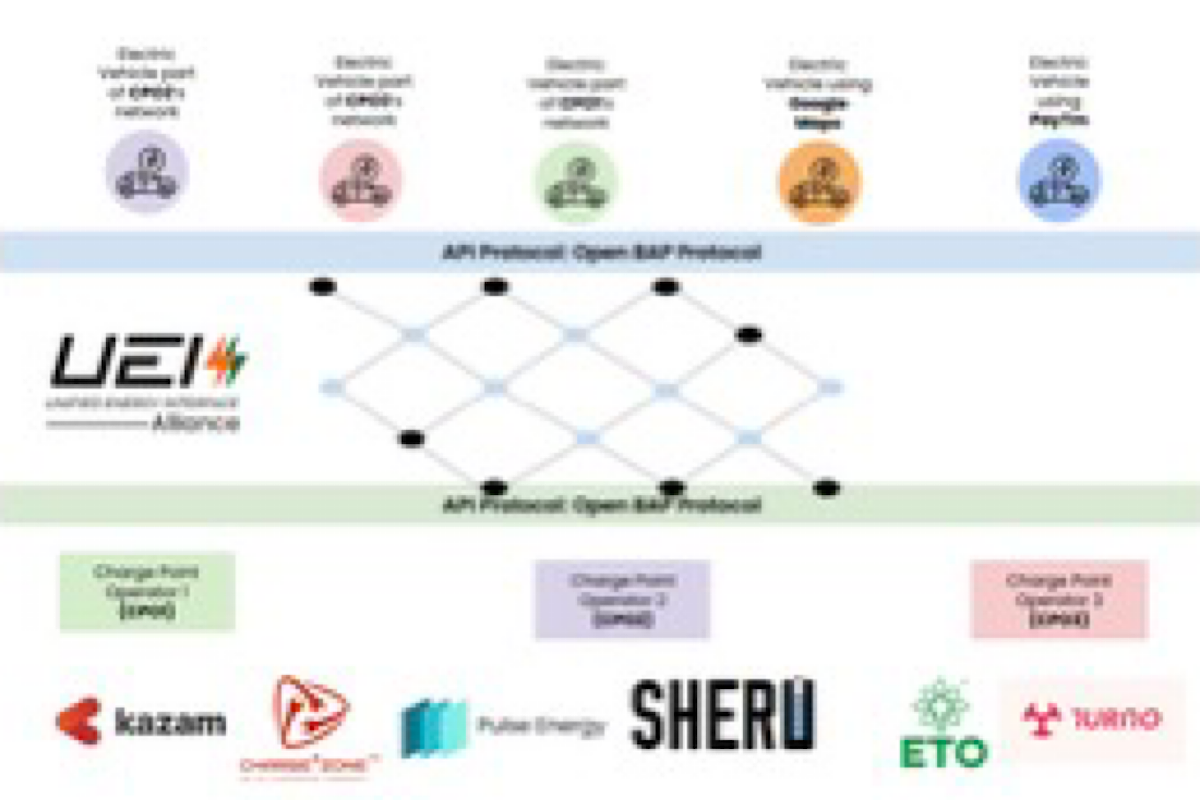
UEI is the recommended interoperability standard as per the Department of Science and Technology.

Chip maker Intel on Friday said that it appointed Santhosh Viswanathan to head the India region business as its Managing Director (MD).

To judge the battery life of any computer, measuring real-world performance like browsing the web, word processing, multimedia editing, etc. is a tough one.
Findings showed that in India, all Cerelac baby products contain an average of nearly 3 grams of sugar per serving.
Semiconductor chips are driving innovation across technology, automotive, telecom, healthcare, and power industries, a new report said on Wednesday.
SAR satellite can create images of photographic maps by emitting electromagnetic waves from space to the ground and synthesising the waves.
Ahead of Elon Musk's visit to India, the tech billionaire's satellite-based internet service Starlink has reportedly received tentative approval from the Ministry of Communications.
If you think you can compensate for your night’s sleep during the daytime you may be wrong, said Dr. Sudhir Kumar, a Hyderabad-based neurologist on Wednesday.